Reactive oxygen species and oxygen
Reactive oxygen species (ROS) are by-products of aerobic metabolism. ROS are involved in both physiological cellular signaling pathways and pathophysiological processes of tissue damage, including brain ischemia-reperfusion injury. The mitochondrial respiratory chain is considered a major source or at least a regulator in the intracellular ROS balance. However, there is little agreement on how ROS production depends on oxygen concentration ([O
2]). We estimated H
2O
2 release by intact mouse brain mitochondria during oxidation of different substrates at various oxygen concentrations. First, we confirmed that the highest rate of H
2O
2 release occurs under conditions of reverse electron transfer (RET).
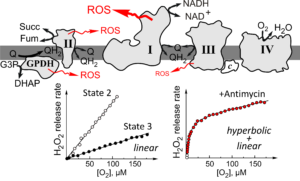
Second, for the first time we determined that H
2O
2 release by respiring mitochondria depends linearly on the oxygen concentration in the physiological range (<200µM [O
2]) during both reverse and forward electron transfer and with any substrate used (malate/pyruvate, succinate, succinate/glutamate or glycerol 3-phosphate). Our results suggest that ROS generation by brain mitochondria depends linearly on oxygen concentration obeying the
law of mass action. H
2O
2 production by Complex III is significant only in the presence of antimycin A, and in this case, the oxygen dependence manifested mixed (linear and hyperbolic) kinetics. The hyperbolic component is contributed by Complex III (Q
o site). The linear dependence of mitochondrial H
2O
2 release on oxygen concentration indicates that ROS production by brain mitochondria in hypoxia is lower than in normoxia.
The very distinctive linear dependence of the H2O2 release on the oxygen concentration is critical for the discussion of the previously proposed hypoxia-stimulated mitochondrial ROS increase, as it is a key event in the regulation of HIF1∝ stabilization.
However, the question still persists: why does antimycin-induced production of ROS by Complex III manifest hyperbolic dependence on oxygen concentration?
Publications
- Stepanova A, Konrad C, Manfredi G, Springett R, Ten V, Galkin A (2019) The dependence of brain mitochondria reactive oxygen species production on oxygen level is linear, except when inhibited by antimycin A. J Neurochem. 148, 731-745, DOI: 10.1111/jnc.14654, PMID: 30582748, PDF
- Stepanova A, Galkin A. (2020) Measurement of mitochondrial H2O2 production under varying O2 tensions. Methods Cell.Biol. 155, 273-293.
- DOI: 10.1016/bs.mcb.2019.12.008 , PMID: 32183962, PDF
- Jain IH, Zazzeron L, Goldberger O, Marutani E, Wojtkiewicz GR, Ast T, Wang H, Schleifer G, Stepanova A, Brepoels K, Schoonjans L, Carmeliet P, Galkin A, Ichinose F, Zapol WM, Mootha VK. (2019) Leigh Syndrome Mouse Model Can Be Rescued by Interventions that Normalize Brain Hyperoxia, but Not HIF Activation. Cell Metab. DOI: 10.1016/j.cmet.2019.07.006, PMID: 31402314, PDF
- Drose S., Galkin A., Brandt, U. (2009). Measurement of superoxide formation by mitochondrial complex I of Yarrowia lipolytica. Methods in Enzymology, 456: 475-490. PMID:19348905, PDF
- Galkin, A. and Brandt, U. (2005) Superoxide radical formation by pure complex I (NADH: Ubiquinone oxidoreductase) from Yarrowia lipolytica. J.Biol.Chem., 280:30129-30135. PMID:15985426, PDF
 Second, for the first time we determined that H2O2 release by respiring mitochondria depends linearly on the oxygen concentration in the physiological range (<200µM [O2]) during both reverse and forward electron transfer and with any substrate used (malate/pyruvate, succinate, succinate/glutamate or glycerol 3-phosphate). Our results suggest that ROS generation by brain mitochondria depends linearly on oxygen concentration obeying the law of mass action. H2O2 production by Complex III is significant only in the presence of antimycin A, and in this case, the oxygen dependence manifested mixed (linear and hyperbolic) kinetics. The hyperbolic component is contributed by Complex III (Qo site). The linear dependence of mitochondrial H2O2 release on oxygen concentration indicates that ROS production by brain mitochondria in hypoxia is lower than in normoxia.
The very distinctive linear dependence of the H2O2 release on the oxygen concentration is critical for the discussion of the previously proposed hypoxia-stimulated mitochondrial ROS increase, as it is a key event in the regulation of HIF1∝ stabilization.
However, the question still persists: why does antimycin-induced production of ROS by Complex III manifest hyperbolic dependence on oxygen concentration?
Second, for the first time we determined that H2O2 release by respiring mitochondria depends linearly on the oxygen concentration in the physiological range (<200µM [O2]) during both reverse and forward electron transfer and with any substrate used (malate/pyruvate, succinate, succinate/glutamate or glycerol 3-phosphate). Our results suggest that ROS generation by brain mitochondria depends linearly on oxygen concentration obeying the law of mass action. H2O2 production by Complex III is significant only in the presence of antimycin A, and in this case, the oxygen dependence manifested mixed (linear and hyperbolic) kinetics. The hyperbolic component is contributed by Complex III (Qo site). The linear dependence of mitochondrial H2O2 release on oxygen concentration indicates that ROS production by brain mitochondria in hypoxia is lower than in normoxia.
The very distinctive linear dependence of the H2O2 release on the oxygen concentration is critical for the discussion of the previously proposed hypoxia-stimulated mitochondrial ROS increase, as it is a key event in the regulation of HIF1∝ stabilization.
However, the question still persists: why does antimycin-induced production of ROS by Complex III manifest hyperbolic dependence on oxygen concentration?